About eighty percent of the periodic table is made up of metals. They make up the majority of the elements and are distinguished from non-metals and semi-metals.
Metals and non-metals in the periodic table
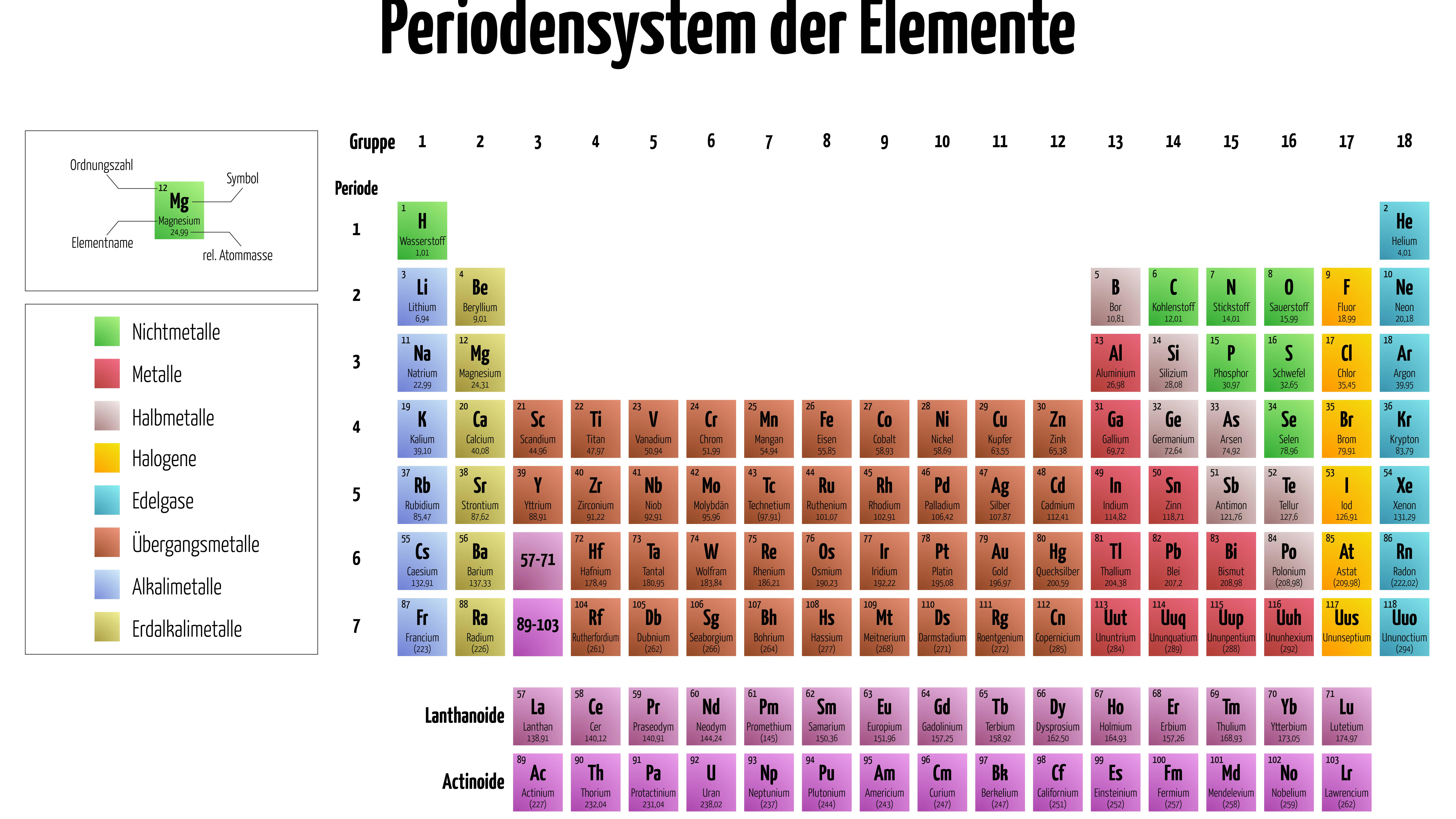
The periodic table is an overview of the known elements. The chemical elements are sorted by similar properties and by nuclear charge.
- Eighty percent of the known chemical elements are metals. Therefore, one criterion for distinguishing between elements is whether they are metal or non-metal. There are also some semi-metals.
- In the periodic table, the metals are on the left and extend to the middle on the right. The non-metals are listed from the right. However, it is not possible to draw a clear dividing line, so there are so-called semi-metals between metals and non-metals.
- Metals share typical properties such as conductivity of electricity or heat, high density and metallic luster. Non-metals, on the other hand, have mostly opposite properties.
This is where the metals are found
The periodic table is divided into eight groups, some of which are dominated by metals.
- Main group 1 contains the alkali metals. These include lithium, sodium, potassium, rubidium, caesium and francium. In the periodic table, the elements are always indicated by abbreviations. In this case, Li, Na, K, Rb, Cs and Fr.
- The second main group contains the alkaline earth metals, which include beryllium, magnesium, calcium, strontium, barium and radium. The third main group is called the boron group, although boron is not a metal. This group includes aluminum, gallium, indium and thallium.
- There are also metals in main group four, the carbon group. These include nitrogen, phosphorus and semi-metals. The fifth main group, called the nitrogen group, also contains the metal bismuth. There are no metals in the other three groups.
You might also be interested in:
- Noble gases and their properties: All information
- Difference between element and compound: Simply explained
- Elementary particles and the atomic structure: The most important information
